St. Luke’s Cancer Center announced this week that it has introduced an innovative pre-treatment option, SpaceOAR Hydrogel, for prostate cancer patients.
SpaceOAR Hydrogel was developed to reduce rectal complications and help prostate cancer patients maintain their quality of life following radiation treatment. It is the only such treatment cleared by the FDA to help reduce the radiation dose delivered to the rectum of men undergoing radiation treatment for prostate cancer.
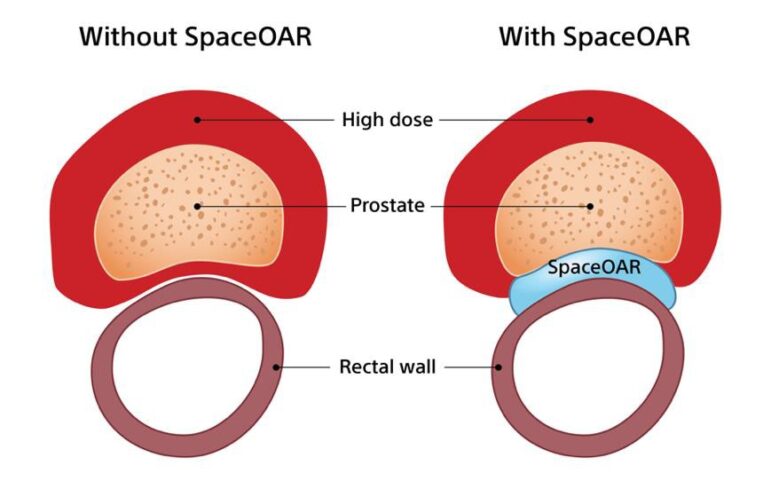
Because of the proximity of the prostate to the rectum, prostate radiation therapy can cause unintended damage to the rectum, which can lead to fecal incontinence issues or other long-lasting side effects.
SpaceOAR Hydrogel is an absorbable gel inserted via a minimally invasive procedure that creates a temporary space between the prostate and the rectum, allowing the doctor to complement the patient’s radiation treatment to better target their cancer while preserving healthy tissue.
Contributed image
The hydrogel spacer remains in place for about three months. After about six months, the hydrogel is naturally absorbed and cleared from the body in the patient’s urine.
“SpaceOAR is an important advance that reduces radiation dose to the rectum during radiation treatments to help prevent unwanted side effects,” said Nimisha Deb, MD, Chief of Radiation Oncology for St. Luke’s. “We understand that men diagnosed with prostate cancer may have some difficult decisions to make, but knowing that an option like SpaceOAR Hydrogel can help limit the risk of damage to other organs during radiation treatment will hopefully make a few of those decisions easier.”
Prostate cancer is the most common cancer in American men, with more than 183,000 new cases diagnosed each year.
Each year more than 60,000 American men opt to treat their prostate cancer with radiation.
In a randomized clinical study, men who underwent radiation therapy without SpaceOAR Hydrogel were eight times more likely to experience a decline in bowel, urinary and sexual function when compared to men who underwent radiation therapy with SpaceOAR Hydrogel, at a median follow-up of three years.
“We are recommending SpaceOAR Hydrogel for many of our prostate cancer patients,” said Richard Lieberman, MD, urologist with St. Luke’s University Health Network. “It’s a very quick and easy procedure to place the gel, and the benefits are tremendous.”
FDA clearance was granted for SpaceOAR Hydrogel following completion of a prospective, multicenter, randomized clinical trial.
The full clinical trial results have been published in the peer-reviewed Red Journal.
Note: This local health news is brought to you in partnership with St. Luke’s University Health Network.







